Transcript
ReachMD Announcer:
This program is brought to you by Regeneron. The speaker is a paid consultant.
Dr. Turck:
Welcome to Eye on Ocular Health on ReachMD. I'm Dr. Charles Turck, and I’m delighted to have you with us as we do a deep dive into EYLEA® (aflibercept) Injection 2 milligrams as a treatment option for Macular Edema following Retinal Vein Occlusion, or RVO.
Once again, we have the pleasure of speaking with Dr. John Kitchens, on behalf of Regeneron Pharmaceuticals. Thank you for continuing this important discussion with us.
Dr. Kitchens:
Thanks, Dr. Turck. I’m looking forward to our discussion today.
Dr. Turck:
Before we begin, let’s review the Indications and some Important Safety Information for EYLEA.
Narrator:
IMPORTANT SAFETY INFORMATION AND INDICATIONS
INDICATIONS
EYLEA® (aflibercept) Injection 2 mg is indicated for the treatment of patients with Neovascular (Wet) Age-Related Macular Degeneration (AMD), Macular Edema following Retinal Vein Occlusion (RVO), Diabetic Macular Edema (DME), and Diabetic Retinopathy (DR).
CONTRAINDICATIONS
EYLEA is contraindicated in patients with ocular or periocular infections, active intraocular inflammation, or known hypersensitivity to aflibercept or to any of the excipients in EYLEA.
Dr. Turck:
Dr. Kitchens, could you please provide an overview of the VIBRANT, COPERNICUS, AND GALILEO pivotal trials?
Dr. Kitchens:
[Slide on screen]

Of course. So let’s start with VIBRANT, which was a multicenter, randomized, double-masked controlled trial in patients with Macular Edema following Branch RVO.1,2
A total of 181 patients with MEfBRVO (or HRVO) were randomly assigned in a 1:1 ratio to receive either EYLEA ® (aflibercept) Injection 2 milligrams every 4 weeks or laser photocoagulation administered at baseline and subsequently as needed.1
Now, laser photocoagulation served as the comparator control arm, being the standard treatment for MEfBRVO at the time this trial was conducted.3
The primary endpoint was the proportion of patients gaining 15 or more Early Treatment Diabetic Retinopathy Study, or ETDRS letters of best-corrected visual acuity, or BCVA at Week 24 from baseline.1 Secondary endpoints included mean change in BCVA, as measured by ETDRS letters, and mean change in central retinal thickness, or CRT at Week 24 from baseline.2
Patients in the EYLEA group received EYLEA 2 milligrams every 4 weeks from baseline through Week 20. These patients also received a sham laser treatment at baseline.2,4
Patients in the EYLEA group who met at least 1 rescue criterion at Weeks 12, 16, or 20 received a sham laser treatment.
Patients in the laser control group received laser photocoagulation at baseline and received 1 additional laser photocoagulation treatment from Week 12 to Week 20 if eligible for rescue treatment, with sham injections every 4 weeks from baseline through Week 20.2,4 The rescue criteria that was applied can be seen on the slide.
[Slide on screen]

Similarly, both COPERNICUS and GALILEO were also randomized, double-masked, multicenter trials, but of patients with Macular Edema following Central Retinal Vein Occlusion. There were 187 patients in COPERNICUS and 171 patients in GALILEO.1
Patients were randomly assigned in a 3:2 ratio to either EYLEA® (aflibercept) Injection 2 milligrams every 4 weeks or to sham intravitreal injections every 4 weeks.1 Panretinal photocoagulation was available to all patients in both studies.5,6
As in the VIBRANT study, the primary endpoint for these studies was the proportion of patients gaining 15 or more letters on the ETDRS scale of best-corrected visual acuity at Week 24 from baseline.1,5,6 Secondary endpoints included mean change in best-corrected visual acuity, as measured by ETDRS letters, and mean change in central retinal thickness at Week 24 from baseline.5,6
Dr. Turck:
Thanks for walking us through the study designs, Dr. Kitchens. I’m interested to learn the outcomes for patients in these clinical trials, but first let’s pause for a moment to review some more Important Safety Information for EYLEA.
Narrator:
WARNINGS AND PRECAUTIONS
Intravitreal injections, including those with EYLEA, have been associated with endophthalmitis and retinal detachments and, more rarely, retinal vasculitis with or without occlusion. Proper aseptic injection technique must always be used when administering EYLEA. Patients and/or caregivers should be instructed to report any signs and/or symptoms suggestive of endophthalmitis, retinal detachment, or retinal vasculitis without delay and should be managed appropriately.
Acute increases in intraocular pressure have been seen within 60 minutes of intravitreal injection, including with EYLEA. Sustained increases in intraocular pressure have also been reported after repeated intravitreal dosing with VEGF inhibitors. Intraocular pressure and the perfusion of the optic nerve head should be monitored and managed appropriately.
Dr. Turck:
We’ve previously discussed the importance of preserving vision for our patients with Macular Edema following RVO. Could you please walk us through the outcomes of the primary endpoint in these studies?
Dr. Kitchens:
Certainly. As previously noted in the study designs for VIBRANT, COPERNICUS, and GALILEO, the primary efficacy endpoint was the proportion of patients who gained ≥15 letters in best-corrected visual acuity at Month 6 compared with baseline.1
[Slide on screen]
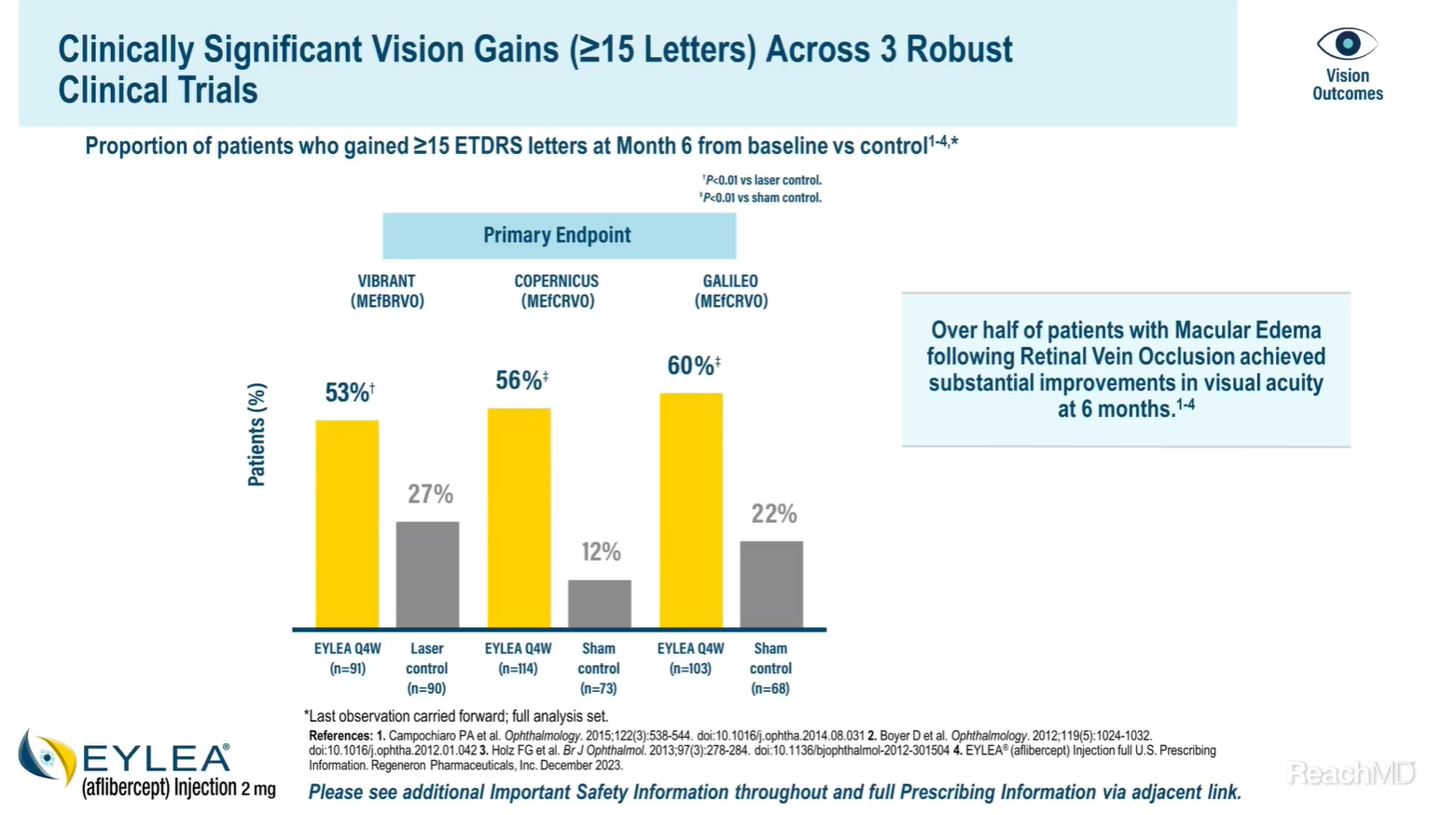
Across these 3 robust clinical trials, more than half of the patients achieved substantial improvements in visual acuity at Month 6.1,2,5,6
In the VIBRANT study, 53% of patients in the EYLEA® (aflibercept) Injection group gained 15 or more ETDRS letters versus 27% in the laser control group at Week 24.1,2
In the COPERNICUS study, 56% of patients in the EYLEA group gained 15 or more ETDRS letters versus 12% in the control group at Week 24.1,5
In the GALILEO study, 60% of patients in the EYLEA group gained 15 or more ETDRS letters versus 22% in the control group at Week 24.1,6
Dr. Turck:
It’s good to hear that more than half of patients treated with EYLEA achieved substantial improvements in visual acuity at Month 6. What about the results for the secondary endpoints?
Dr. Kitchens:
[Slide on screen]
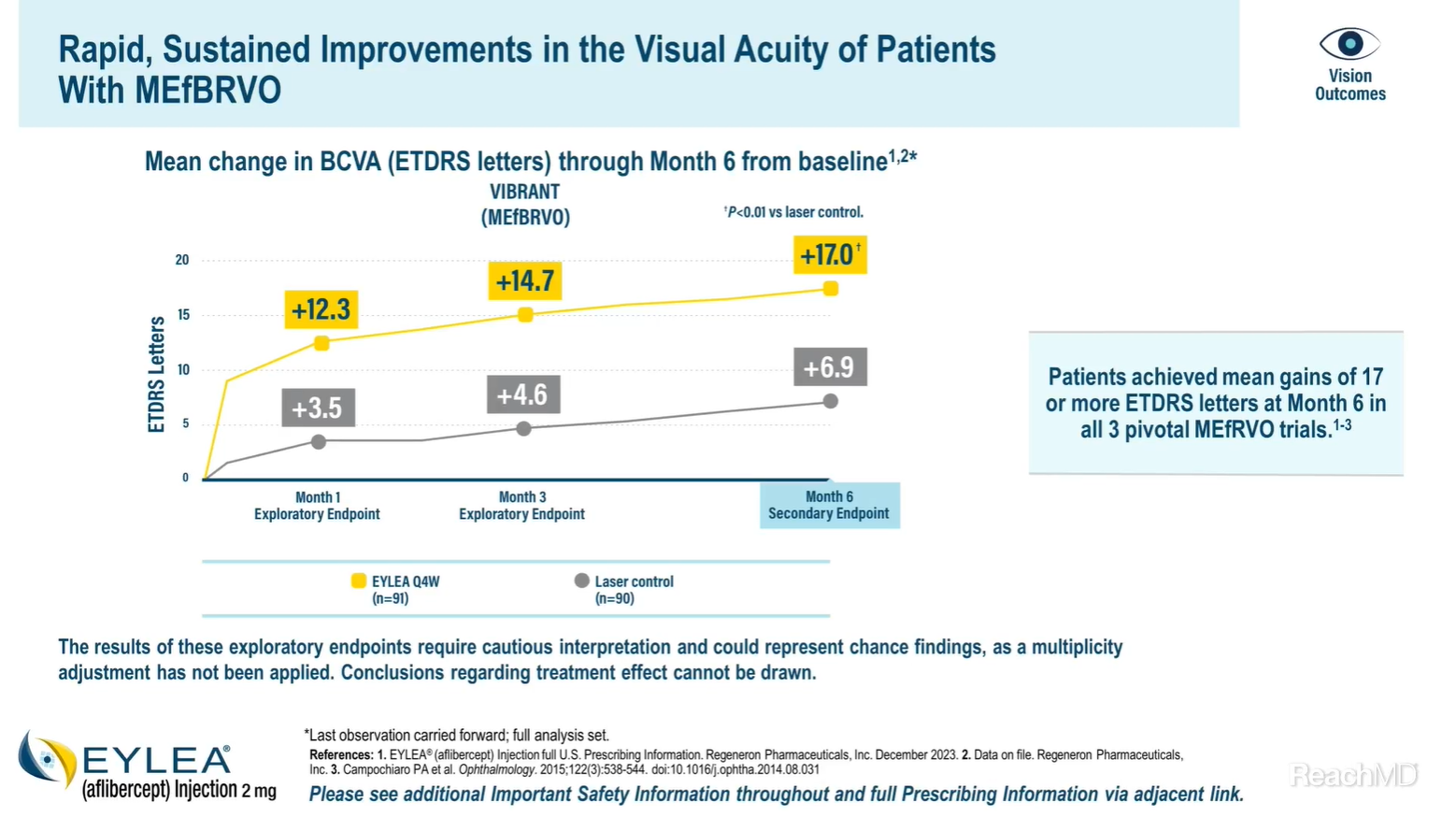
Well, as with the primary endpoint, the secondary endpoints in the pivotal trials were met as well.2,5,6 The mean change in best-corrected visual acuity, as measured by ETDRS letters, from baseline at Month 6 was a prespecified secondary endpoint of the VIBRANT trial.2
EYLEA® (aflibercept) Injection led to rapid and sustained improvements in visual acuity. At Month 1, the mean change in best-corrected visual acuity from baseline was 12.3 letters in the EYLEA group versus 3.5 letters in the laser control group. This gain was maintained through Month 3 and Month 6. At Month 6, the mean change in best-corrected visual acuity from baseline was significantly higher in the EYLEA group vs the laser control group (17.0 vs 6.9 letters, respectively).2,7
Please note that the results of these exploratory endpoints at Months 1 and 3 require cautious interpretation and could represent chance findings, as a multiplicity adjustment has not been applied. Therefore, conclusions regarding treatment effect really can't be drawn from this Months 1 and Months 3.
[Slide on screen]
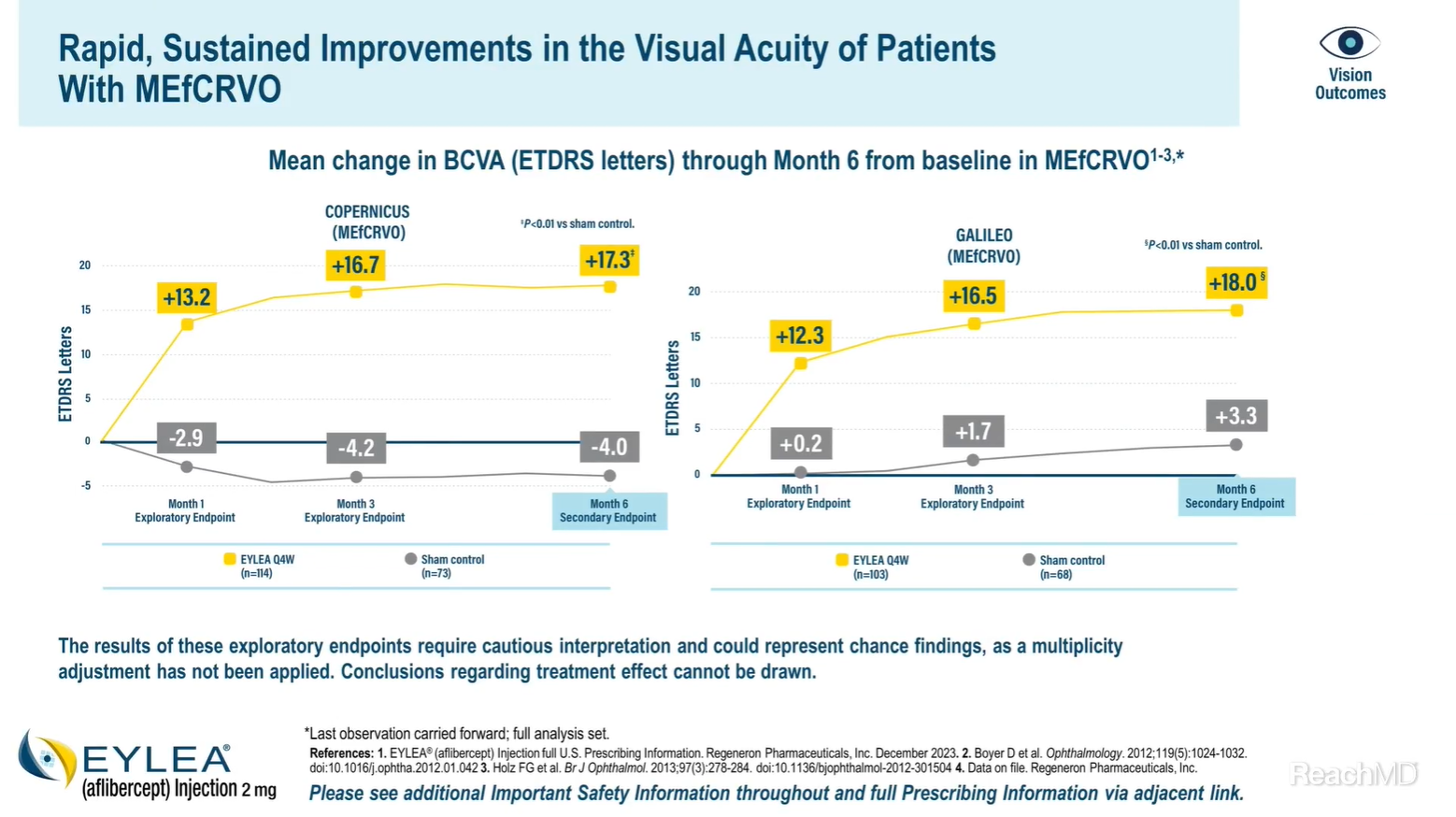
In the COPERNICUS study, the mean change in best-corrected visual acuity from baseline was significantly higher in the EYLEA group versus the control group at Month 6 (17.3 vs -4.0 letters, respectively).5
In the GALILEO study, the mean change in best-corrected visual acuity from baseline was significantly higher in the EYLEA group versus the control group at Month 6 (18.0 vs 3.3 letters, respectively.)6
Dr. Turck:
Thank you, Dr. Kitchens. So, to reiterate, across all 3 pivotal trials, EYLEA led to mean gains of 17 or more ETDRS letters from baseline at Month 6.1 Did baseline visual acuity influence these outcomes?
Dr. Kitchens:
That’s a great question. EYLEA resulted in efficacy across a wide range of patient types in Macular Edema following RVO.
[Slide on screen]
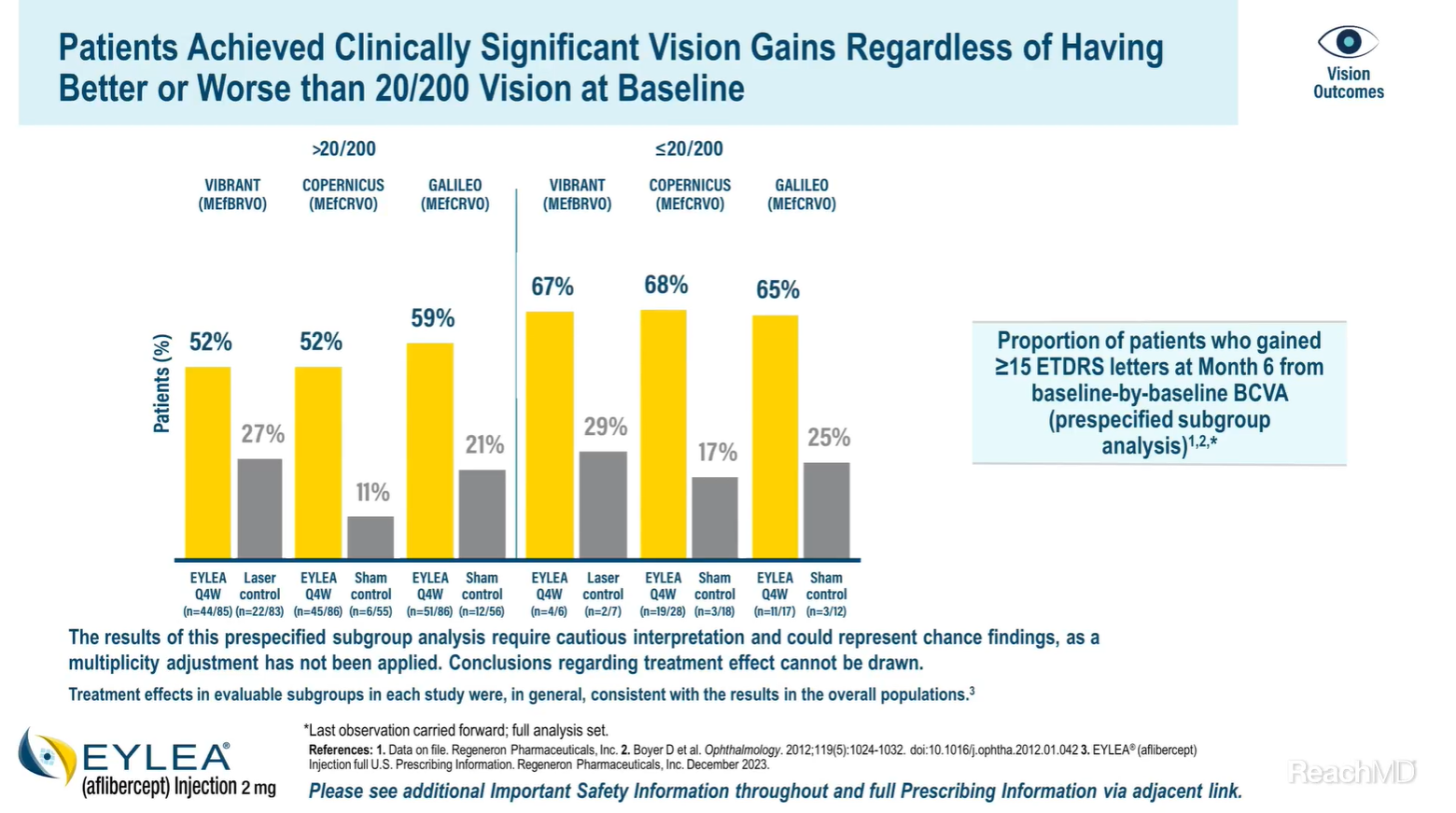
Whether patients’ vision at baseline was better or worse than 20/200, the majority of patients achieved clinically significant vision gains with EYLEA® (aflibercept) Injection.7
In the VIBRANT trial, 52% versus 27% of patients with a visual acuity better than 20/200 at baseline gained ≥15 letters in the EYLEA group versus the laser control group, respectively. In patients with a visual acuity of 20/200 or worse at baseline, 67% in the EYLEA group and 29% in the laser control group gained ≥15 letters.7
In the COPERNICUS trial, 52% versus 11% of patients with a visual acuity better than 20/200 at baseline gained ≥15 letters in the EYLEA group versus the control group, respectively. In patients with a visual acuity of 20/200 or worse at baseline, 68% in the EYLEA group and 17% in the control group gained ≥15 letters.7
In the GALILEO trial, 59% versus 21% of patients with a visual acuity better than 20/200 at baseline gained 15 letters or more in the EYLEA group versus the control group, respectively. In patients with a visual acuity of 20/200 or worse at baseline, 65% in the EYLEA group and 25% in the control group gained ≥15 letters.7
It's important to note that the results of this prespecified subgroup analysis require cautious interpretation and could represent chance findings, as a multiplicity adjustment has not been applied. Conclusions regarding treatment effect cannot be drawn.
The treatment effects in the evaluable subgroups in each study were, in general, consistent with the results in the overall population.1
Dr. Turck:
So, patients benefited from EYLEA 2 mg treatment regardless of whether their vision at baseline was better or worse than 20/200. That’s a nice segue into my next question: Did timing of when the EYLEA intervention occurred impact the amount of vision that was gained?
Dr. Kitchens:
[Slide on screen]
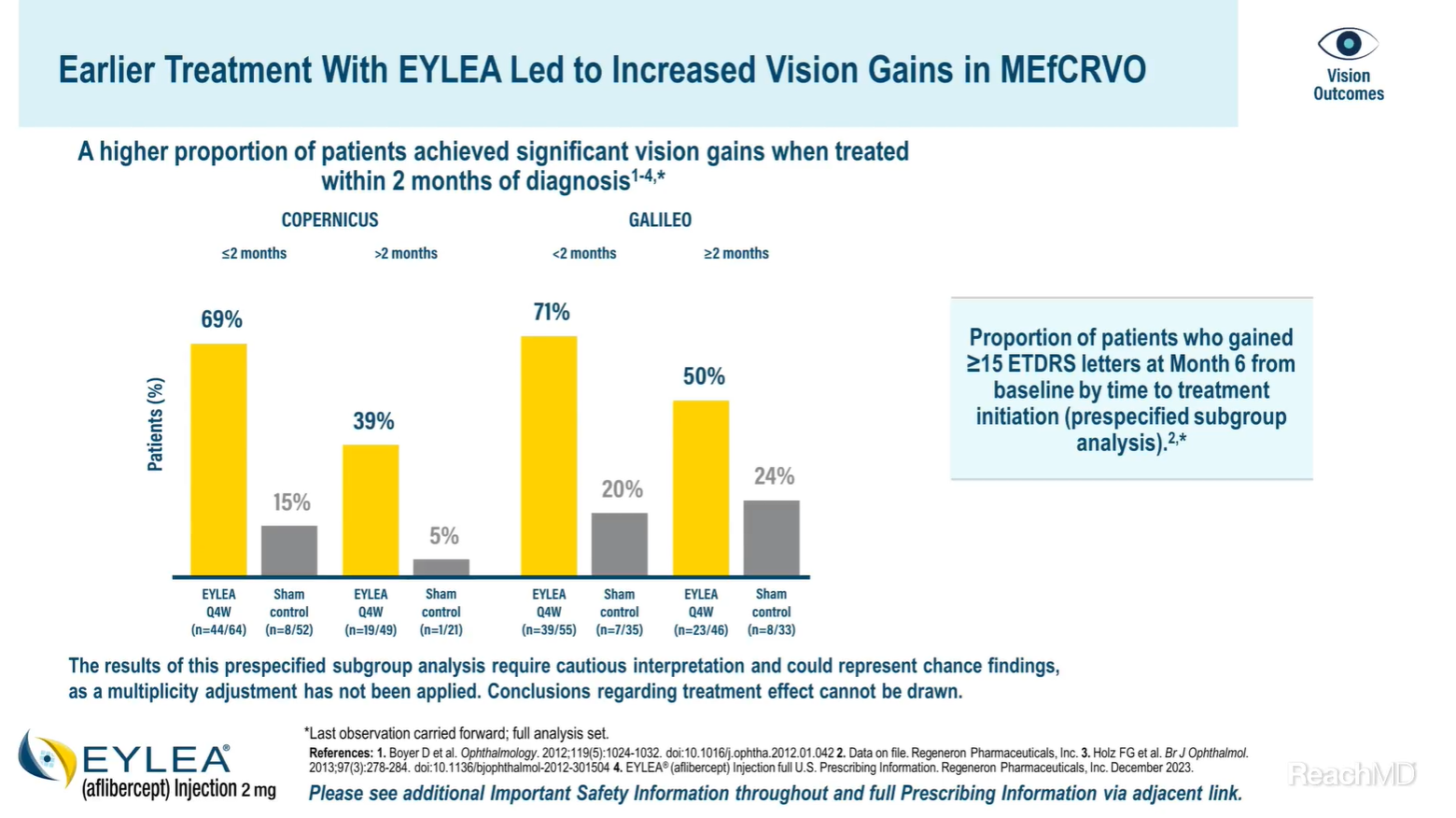
Yes, in both the COPERNICUS and GALILEO trials, early treatment with EYLEA® (aflibercept) Injection in macular edema following central retinal vein occlusion resulted in greater vision gains versus controls.7
In COPERNICUS, 68.8% of patients treated with EYLEA within 2 months of diagnosis gained 15 or more letters of vision versus 15.4% in the control group at Month 6 from baseline. In patients in whom treatment was initiated at greater than 2 months after diagnosis, 38.8% receiving EYLEA and 4.8% in the control group gained 15 letters of vision or more at Month 6 from baseline.7
In GALILEO, 70.9% of patients treated with EYLEA within 2 months of diagnosis gained ≥15 letters of vision versus 20.0% in the control group at Month 6. In patients in whom treatment was initiated at greater than 2 months after diagnosis, 50.0% receiving EYLEA and 24.2% in the control group gained ≥15 letters of vision at Month 6.7
As we previously noted just a moment ago, these data also require cautious interpretation and could represent chance findings.
Dr. Turck:
Thanks, Dr. Kitchens. So, a greater proportion of patients achieved substantial vision gains when treated within 2 months of diagnosis in the COPERNICUS and GALILEO trials.7 Before we move on, let’s pause again to continue reviewing some Important Safety Information on EYLEA.
Narrator:
WARNINGS AND PRECAUTIONS (continued)
There is a potential risk of arterial thromboembolic events (ATEs) following intravitreal use of VEGF inhibitors, including EYLEA. ATEs are defined as nonfatal stroke, nonfatal myocardial infarction, or vascular death (including deaths of unknown cause). The incidence of reported thromboembolic events in wet AMD studies during the first year was 1.8% (32 out of 1824) in the combined group of patients treated with EYLEA compared with 1.5% (9 out of 595) in patients treated with ranibizumab; through 96 weeks, the incidence was 3.3% (60 out of 1824) in the EYLEA group compared with 3.2% (19 out of 595) in the ranibizumab group. The incidence in the DME studies from baseline to week 52 was 3.3% (19 out of 578) in the combined group of patients treated with EYLEA compared with 2.8% (8 out of 287) in the control group; from baseline to week 100, the incidence was 6.4% (37 out of 578) in the combined group of patients treated with EYLEA compared with 4.2% (12 out of 287) in the control group. There were no reported thromboembolic events in the patients treated with EYLEA in the first six months of the RVO studies.
Dr. Turck:
Dr. Kitchens, we have discussed vision gains which were achieved with EYLEA regardless of baseline visual acuity. What were the anatomic outcomes in these patients?
Dr. Kitchens:
[Slide on screen]
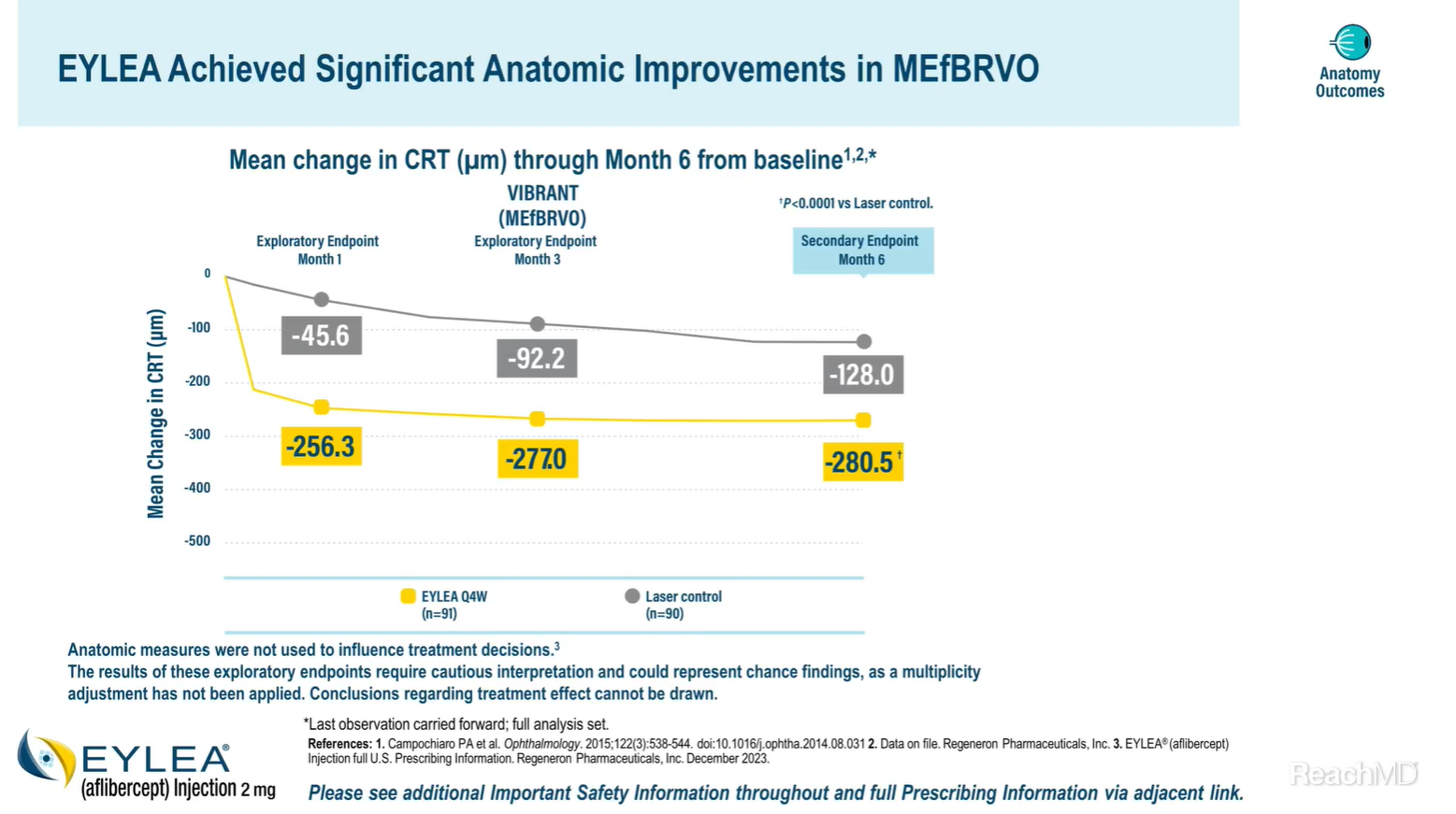
Well, Dr. Turck, as with vision improvements, anatomic improvements in macular edema following branch retinal vein occlusion were also demonstrated with EYLEA® (aflibercept) Injection. Mean change from baseline in central retinal thickness at Month 6 was a prespecified secondary endpoint of the VIBRANT trial.2
Treatment with EYLEA resulted in reductions in CRT compared with the laser control group through Month 6. The mean reduction in CRT from baseline was 280.5 versus 128.0 µm at Month 6 in the EYLEA group versus the laser control group, respectively.2
Anatomic data in this trial were not used to influence treatment decisions.1
[Slide on screen]
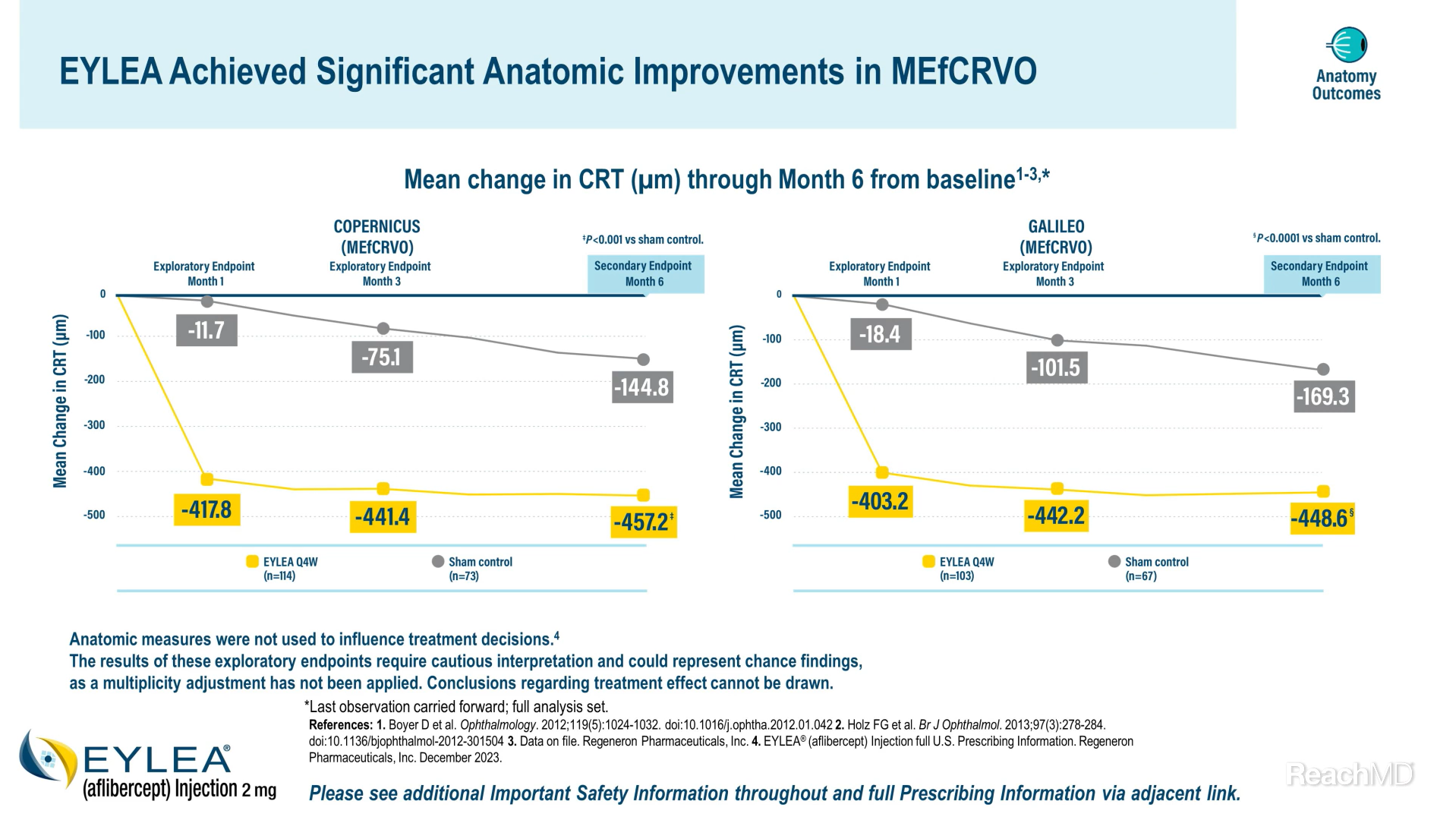
And as in Macular Edema following Branch Retinal Vein Occlusion, anatomic improvements with EYLEA were also demonstrated in Macular Edema following Central Retinal Vein Occlusion. Mean change from baseline in CRT at Month 6 was a prespecified secondary endpoint of both the COPERNICUS and GALILEO trials.5,6
In COPERNICUS, the mean reduction in CRT from baseline was 457.2 versus 144.8 µm at Month 6 in the EYLEA group versus the sham control group, respectively.5
In GALILEO, the mean reduction in CRT from baseline was 448.6 versus 169.3 µm at Month 6 in the EYLEA group versus the sham control group, respectively.6
Dr. Turck:
Thank you, Dr. Kitchens. And now, let’s finish reviewing the Important Safety Information.
Narrator:
ADVERSE REACTIONS
Serious adverse reactions related to the injection procedure have occurred in <0.1% of intravitreal injections with EYLEA® (aflibercept) Injection including endophthalmitis and retinal detachment.
The most common adverse reactions (≥5%) reported in patients receiving EYLEA were conjunctival hemorrhage, eye pain, cataract, vitreous detachment, vitreous floaters, and intraocular pressure increased.
Patients may experience temporary visual disturbances after an intravitreal injection with EYLEA and the associated eye examinations. Advise patients not to drive or use machinery until visual function has recovered sufficiently.
Dr. Turck:
Now, Dr. Kitchens, you had mentioned previously that patients with Macular Edema following RVO are generally more likely to have certain underlying medical conditions or comorbidities, and as always it is important for us to consider the safety profile of a selected treatment. Can you discuss the safety data of EYLEA from these trials?
Dr. Kitchens:
[Slide on screen]
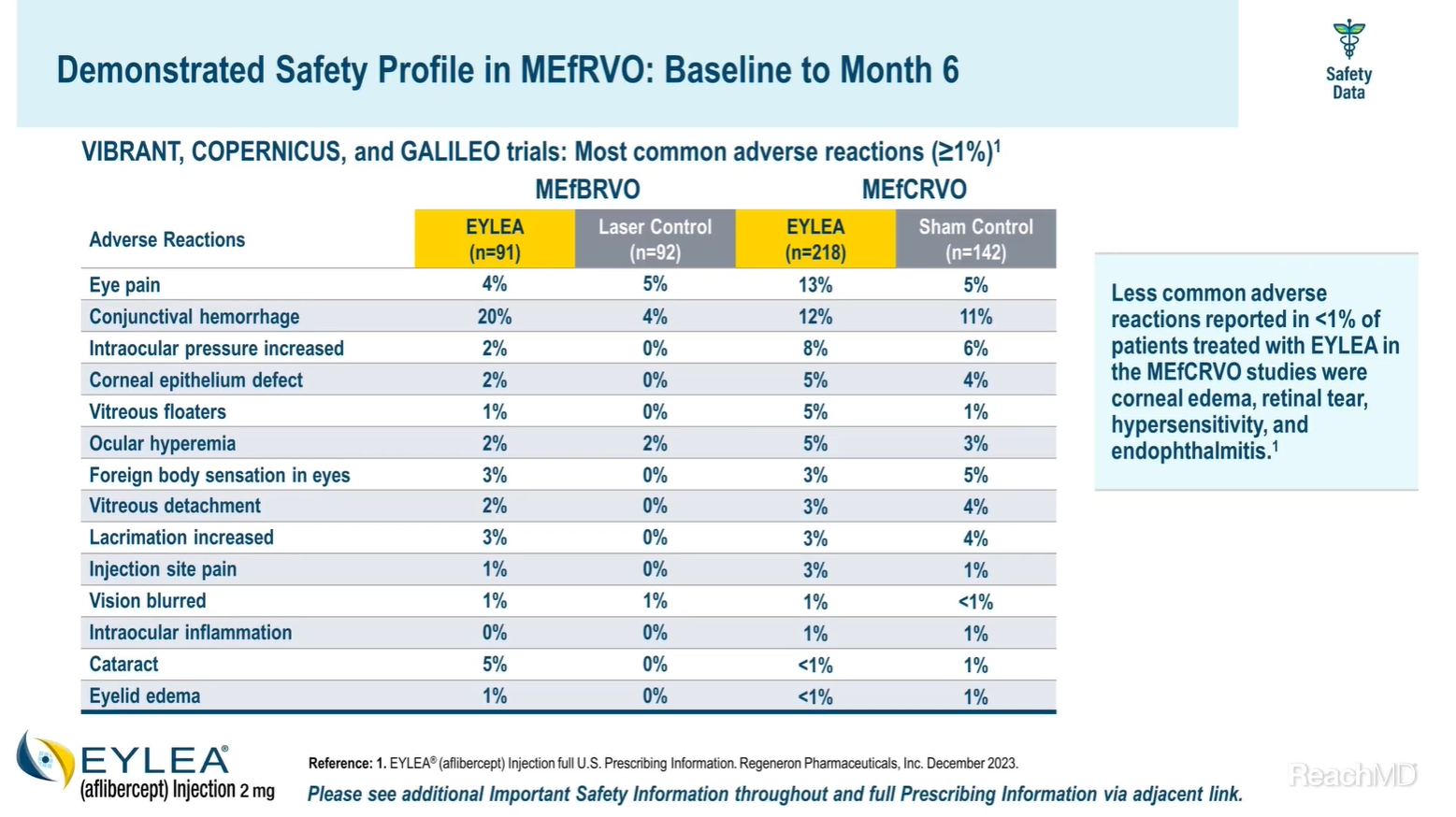
Yes, of course, EYLEA had a demonstrated safety profile in Macular Edema following Retinal Vein Occlusion across all 3 trials. The most common adverse reactions (occurring in at least 5% of patients) reported in patients with macular edema following branch retinal vein occlusion and receiving EYLEA were conjunctival hemorrhage and cataract, whereas the most common adverse reactions (at least 5%) reported in patients with macular edema following central retinal vein occlusion and receiving EYLEA were eye pain, conjunctival hemorrhage, intraocular pressure increased, corneal epithelium defect, vitreous floaters, and ocular hyperemia.1
Less common adverse reactions reported in less than 1% of patients treated with EYLEA in the macular edema following central retinal vein occlusion studies were corneal edema, retinal tear, hypersensitivity, and endophthalmitis.1
[Slide on screen]
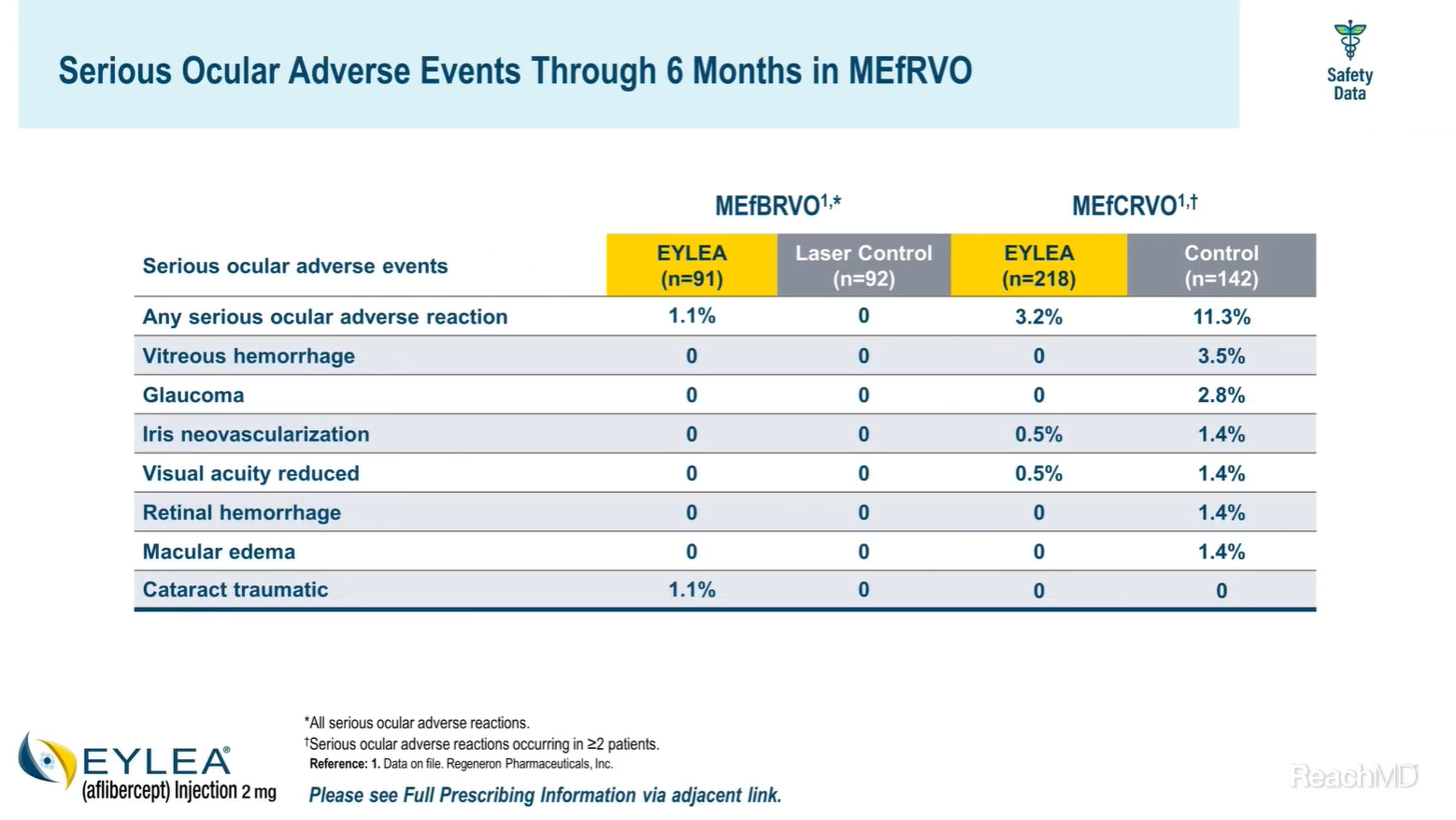
One serious ocular adverse event, traumatic cataract, was reported in the VIBRANT study. In the COPERNICUS and GALILEO studies, serious adverse events were reported in 3.2% of eyes receiving EYLEA® (aflibercept) Injection and 11.3% of eyes receiving control.5-7
There were no incidents of serious treatment-emergent vitreous or retinal hemorrhage in the EYLEA arms across all RVO trials through Month 6.1
Dr. Turck:
Thank you, Dr. Kitchens, here on behalf of Regeneron Pharmaceuticals, for helping us better understand the efficacy and safety data for EYLEA in patients with Macular Edema following RVO across these 3 clinical trials that remain relevant today.
Dr. Kitchens:
Thanks for having me.
ReachMD Announcer:
This program is brought to you by Regeneron. The speaker is a paid consultant.
References:
- EYLEA® (aflibercept) Injection full U.S. Prescribing Information. Regeneron Pharmaceuticals, Inc. December 2023.
- Campochiaro PA, Clark WL, Boyer DS, et al. Intravitreal aflibercept for macular edema following branch retinal vein occlusion: the 24-week results of the VIBRANT study. Ophthalmology. 2015;122(3):538-544.
- Panakanti TK, Chhablani J. Clinical trials in branch retinal vein occlusion. Middle East Afr J Ophthalmol. 2016;23(1):38-43.
- Clark WL, Boyer DS, Heier JS, et al. Intravitreal aflibercept for macular edema following branch retinal vein occlusion: 52-week results of the VIBRANT study. Ophthalmology. 2016;123(2):330-336.
- Boyer D, Heier J, Brown DM, et al. Vascular endothelial growth factor trap-eye for macular edema secondary to central retinal vein occlusion: six-month results of the phase 3 COPERNICUS study. Ophthalmology. 2012;119(5):1024-1032.
- Holz FG, Roider J, Ogura Y, et al. VEGF trap-eye for macular oedema secondary to central retinal vein occlusion: 6-month results of the phase III GALILEO study. Br J Ophthalmol. 2013;97(3):278-284.
- Data on file. Regeneron Pharmaceuticals, Inc.
© 2024, Regeneron Pharmaceuticals, Inc. All rights reserved.
US.EYL.24.07.0005 08/2024














