Diabetes is one of the leading causes of vision loss in the United States and disproportionately affects racial and ethnic minorities. Our panel of experts in ophthalmology, optometry, and primary care discuss the latest tools they’re using to help their most vulnerable patients. Join us in tackling the barriers to better vision care so that all patients receive the care and treatment they need.
Diabetic retinopathy (DR) and its related diabetic macular edema (DME) are leading causes of vision impairment.1 In the United States (US), approximately 1 of every 25 people aged 40 years and older with diabetes has DME in at least 1 eye.2 Racial and ethnic minorities (ie, Black, Hispanic/Latinx, American Indian) are disproportionately affected and have a 2.3 times greater likelihood of developing visual complications from diabetes than their White counterparts.3 Additionally, individuals impacted by health disparities are at a greater risk of vision loss due to a number of patient, physician, and institutional barriers resulting in suboptimal care.3,4 There is an urgent need for healthcare providers to implement strategies to overcome these barriers and improve outcomes. The following is a roundtable discussion between experts in the fields of ophthalmology, optometry, and family medicine. Hear how they are approaching patient care and find out what tools are available to help patients receive the care and treatment they need.
Background
Charles Vega, MD: Latinx populations have the highest rates of diabetes-related vision loss, particularly due to retinopathy (Figure 1).5,6 They are closely followed by Black populations. We know that diabetes is more common across racial and ethnic minorities (Figure 2).7 I consider those individuals to be at high risk of vision loss, primarily due to a lack of healthcare access.
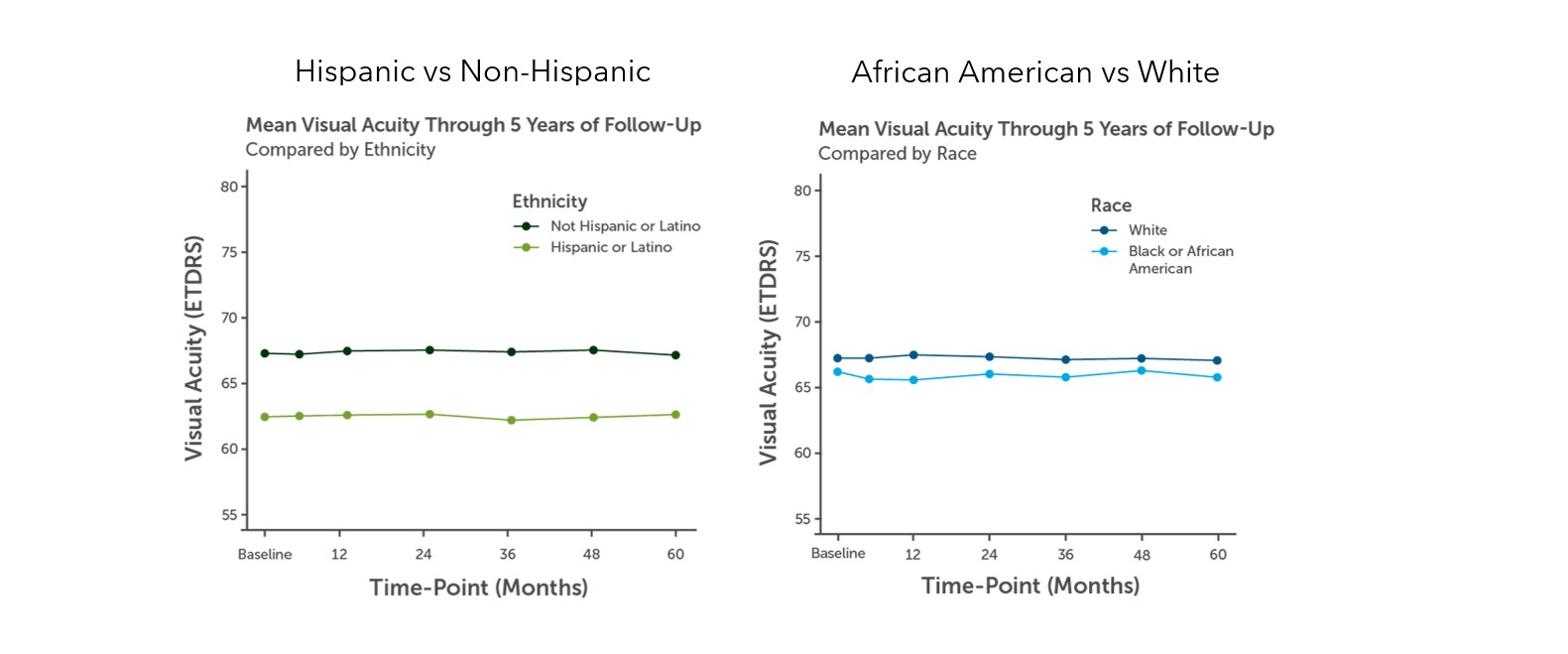
Figure 1. Comparison of visual acuity of patients with DME. (A) Hispanic (B) Black or African American.6
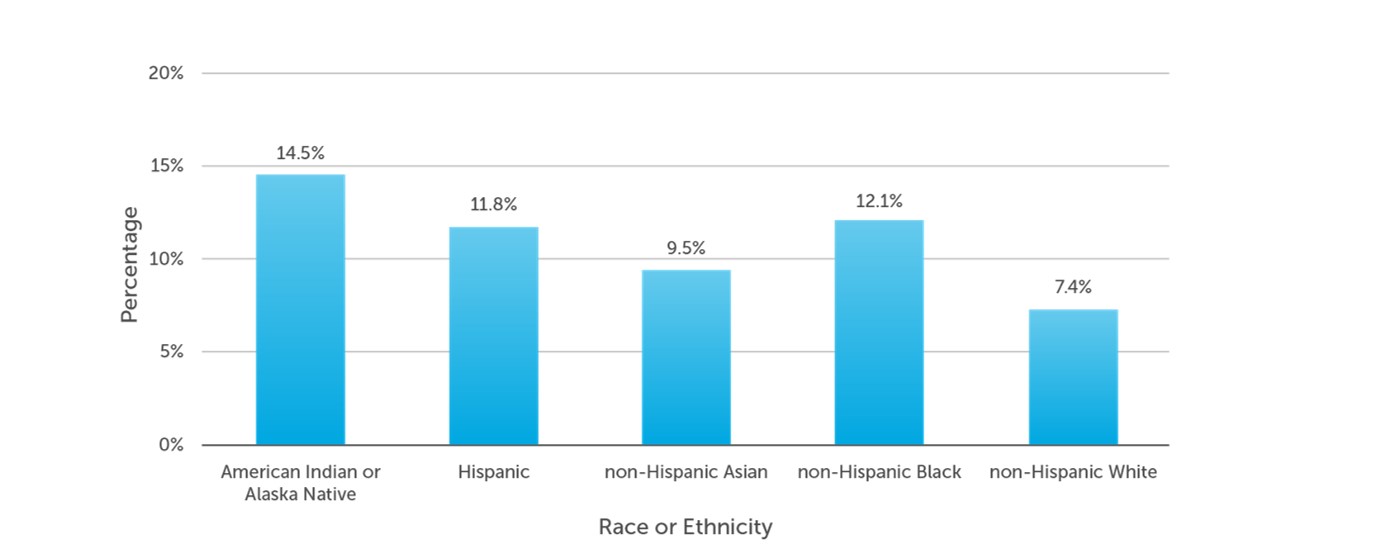
Figure 2. Adults with diabetes by racial or ethnic group.7
It starts with us in primary care making referrals to ophthalmology and making sure those visits are completed. Primary care providers also need to make sure the patient’s diabetes is well controlled along with other factors that can affect vision, such as hypertension and cholesterol. We have to be a team since over one-fifth of individuals with a new diagnosis of diabetes already have retinopathy present.8
Differences in Risk
Pradeep Prasad, MD, MBA: The length of time that one has diabetes is a factor in how likely one is to develop diabetic eye disease.8 In the Los Angeles County clinic where I work, we are seeing a lot more patients with young-onset type 2 diabetes (T2D). We did a study looking at these patients and found that they were at much higher risk of developing end-stage diabetic eye disease, as well as other systemic complications from diabetes, compared to patients with adult-onset T2D who were diabetic for the same amount of time. This younger population with T2D may be a higher-risk group that we need to pay attention to.
Sharon Solomon, MD: Did you find that those patients with young-onset T2D had higher rates of obesity?
Pradeep Prasad, MD, MBA: Obesity was a factor along with other medical comorbidities, like hypertension and hyperlipidemia. There are a whole constellation of factors that may increase risk for this particular group. In our county hospital, the Latino population makes up a huge proportion of our patients. I think ethnic differences are important, but age might also be something that we should pay attention to.
.jpg)
Figure 3. Good management of diabetes requires systemic control of HbA1c, blood pressure, and cholesterol.
Sharon Solomon, MD: It seems that ethnic groups of non-European descent may have more of a genetic predisposition to developing diabetes.
Ruth Shoge, OD, MPH: There is more to the story than genetic predisposition. Quite a few studies have looked at environmental factors, such as stress, as a culprit in disease risk and health outcomes.9 An environment of ongoing toxic stress can lead to the behavioral choices one makes to cope with stress, for example, smoking, drinking, over/under eating, or recreational drug use. This then, of course, increases the risk of diabetes, high blood pressure, heart disease, etc. Even when one lives in pristine health, research—particularly maternal and child health research—has demonstrated the stress of simply existing as a marginalized person plays a role in health outcomes. This ongoing exposure to and living with stress has been coined "weathering."10 Emerging research is also specifically looking at the way epigenetics—how the environment and subsequent behavioral changes can affect the way your genes work—plays a role not only in an individual but also in their descendants as “transgenerational trauma."11 So while genetics may be a compelling component, the environmental stress factor can light the fuse.

Sharon Solomon, MD: Speaking to your point, we also must recognize that low income and rural populations, where there is less access to care, are at risk. As an ophthalmologist and retina specialist, the first thing that I specifically try to discuss with the patient is management of his or her systemic disease (Figure 3).
Ruth Shoge, OD, MPH: Yes, social determinants of health is a necessary conversation that we need to have with our patients to understand their needs and circumstances within which they live, and also with our colleagues, institutions, and policymakers to acknowledge, understand, and address how these determinants impact individual and population health.
New Ways for Patients to Access Vision Care
Charles Vega, MD: All of this speaks to the need to get the patient referred promptly. We should be screening for diabetes in at-risk populations—if the patient is overweight and particularly if they are a person of color. The US Preventive Services Task Force just lowered their screening guidelines down to just being overweight.12 We should be screening over half of the US adult population and promptly sending patients for a dilated eye exam, even if they are in adolescence.
Pradeep Prasad, MD, MBA: That is a huge part of what we try to do is just get the patient into the clinic. In the Los Angeles County Department of Health Services, we have a teleretinal screening program that is really critical to identifying patients who are at risk for vision-threatening disease. Also, when you think of at-risk populations, we must do a good job as providers of getting to where the patients are. That could include community outreach at community fairs. University of California Los Angeles, for example, has a mobile eye clinic to promote outreach efforts. We need to get the message out to communities who may not be aware that a disease could cause blindness and make them aware of therapeutic options or interventions that can prevent or treat diabetic eye disease should it arise.
Sharon Solomon, MD: One thing that is important is to have facilities where patients can access care easily, such as community health centers. Right now, optometric and ophthalmologic care are really lacking in these centers. As many as 70% of community health centers do not provide eye care, so there is no good way to meet the needs of patients with diabetes who are at risk for DR.13

Pradeep Prasad, MD, MBA: The eye clinic in the LA County hospital is one of the busiest clinics in the entire hospital, which speaks to your point that there is a lack of options for patients in the community to access good eye care. The answers to that particular issue are complex, and we certainly have to do a lot in terms of increasing the availability of eye care services. We also need to partner with other ophthalmologists, optometrists, nurse practitioners, physician assistants, nursing staff, and other care extenders to get the message to our patients that if they are diabetic, then they need to get screened for diabetic eye disease.
Charles Vega, MD: This is more anecdotal, but my patients generally believe in the importance of regular eye examinations. I do not feel like I have to sell the concept of getting their eyes screened. The reason is that many of my patients have an aunt or neighbor who has significant vision loss related to diabetes. Compared to some of the other challenges I have in my practice, when it comes to getting their eye screening, patients are really good about getting that done. Even when they face barriers such as no childcare, time off from work, they find a way to get screening done. I also credit the eye care community for bending over backwards by providing weekend and evening clinic hours. That makes a difference. I agree that using technology is necessary if we want to screen millions of patients on a routine basis.
Pradeep Prasad, MD, MBA: Our teleretinal screening program is primarily in the primary care setting. These are non-mydriatic photos that are taken by nursing staff in primary care clinics. The images are then read by trained image readers. Patients who meet certain criteria are then sent to eye clinics for further evaluation. This lowers the barrier in that if the patient is seeing Dr. Vega for their annual well visit, then it is just an easy walk down the hall to get a photo taken. Those types of strategies are what we can use to screen as many people as possible. Even with that system in place, our screening rates for all empaneled patients in LA County is about 60%. So only 60% of patients that we should be screening are getting screened. I don’t have the answers for how we get to 100%, but we certainly need to do more.
Challenges with Patient Follow-up
Sharon Solomon, MD: You both may be in areas where technology adoption is occurring more rapidly than in other parts of the country. My experience is that it is hard to get patients to buy in even when they have levels of DR that are vision threatening. Because the patient has not lost any level of vision, I find that it is hard to get them to follow up and accept the gravity of the situation. I applaud your efforts, but I wonder if you are in areas where technology is utilized more for screening than in other parts of the country. When you say your patients have relatives who have lost vision, that has not typically been my experience either. For some patients, you have to put extra effort into encouraging them to come back for their intravitreal injection because even though they see the changes in the retina on optical coherence tomography imaging, they are not necessarily seeing a difference in vision, so I find that still to be a challenge.
Pradeep Prasad, MD, MBA: There are all sorts of different factors that may play into whether patients follow up or not. Even if they want to, sometimes there may be external factors that preclude them from being able to follow up. We mentioned getting family members involved and educating them so they can play a role in terms of getting patients to show up for their first visit and stay engaged with ongoing treatments.
Charles Vega, MD: There is likely a role for healthcare coordinators to help overcome some of these barriers, particularly if the workers themselves have diabetes. That is something we have leaned on in the past with our diabetic group visits. Those are pretty powerful, to have actual patients motivate other patients and problem-solve with them.
Sharon Solomon, MD: One thing I wanted to ask you, Dr. Vega, since you mentioned that you do not struggle to get patients to come to their initial visit, is do you think there is a trust factor that is built with patients coming into an office where staff look like them and share the same language? Does that help with improving not just adherence but also the health of the patient?
Charles Vega, MD: As Director of UCI’s [University of California Irvine’s] Program in Medical Education for the Latino Community, I do believe that it is important to have greater diversity among medical professionals. However, we cannot wait for the workforce to change. I think in terms of cross-cultural communications, humility is great, and what patients really value most is trust.
Adapting Treatment Plans to Meet Patient Needs
Sharon Solomon, MD: I find there is a cohort of patients who are engaged and understand how important follow-up is, but when it comes time for treatment, patients do not have adequate insurance to get the proper care. For patients with DME, authorization of traditional focal laser therapy may be granted, whereas treatment with anti-vascular endothelial growth factor (VEGF) therapy is not. In academic institutions, sometimes grants are available that will provide patients with free intravitreal therapy. However, the need for therapy is greater than what these grants can support.
Pradeep Prasad, MD, MBA: Adequate insurance coverage is a real problem.One thing that can help is patient assistance programs that can cover the cost of the medication. I think one of the things we need to think about is how there are therapeutic options where there is less of a treatment burden.
Charles Vega, MD: That is a really good point. Make it easier for the patient, the system, and the clinics.
Sharon Solomon, MD: Therapies that have longer duration are combination anti-VEGF and anti-angiopoietin-2 (Ang-2, Figure 4). A working population who has difficulty with repeatedly missing work or relies on someone for transportation would benefit from a more durable treatment since there is less follow-up.
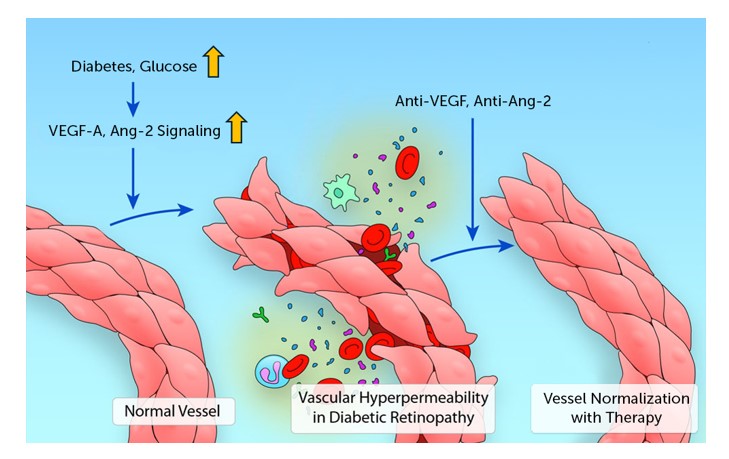
Figure 4. Faricimab is a bispecific inhibitor of VEGF-A and Ang-2. Figure adapted from Claesson-Welsh L. Arterioscler Thromb Vasc Biol. 2018;38(8):1673-1675.
Pradeep Prasad, MD, MBA: Studies indicate that patients treated with faricimab can go 12 to 16 weeks between treatments compared to aflibercept given every 8 weeks (Figure 5).14 Currently, I am using faricimab in patients with whom I am unable to extend their treatment interval or to see if I might be able to get a better therapeutic outcome. Some of my patients are either able to get more effective drying of the macula or longer intervals between treatments. With faricimab and potentially high-dose aflibercept, it is nice to have options. The more therapeutic options we have, the more capable we are going to be to achieve the goal of decreased treatment burden. Currently, we do not know a lot about whether certain patients may respond better to one therapeutic agent than another. I think there is still a lot left to learn, but more treatments mean more options to meet our patients’ needs.
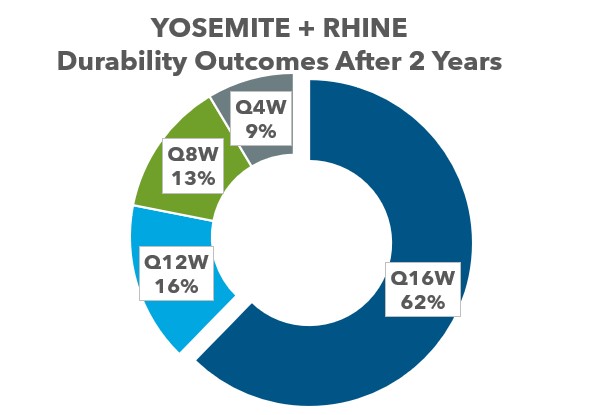
Figure 5. Durability of faricimab in DME after 2 years.14
Sharon Solomon, MD: There are some patients who seem to be truly resistant to anti-VEGF, and it will be encouraging to see if we can get them into remission with faricimab (Figure 6).15 I will use a dexamethasone implant in patients who have had cataract surgery who have not had a good response to anti-VEGF therapy. In the future, the port delivery system (PDS) with ranibizumab may be an option for these patients, as well.
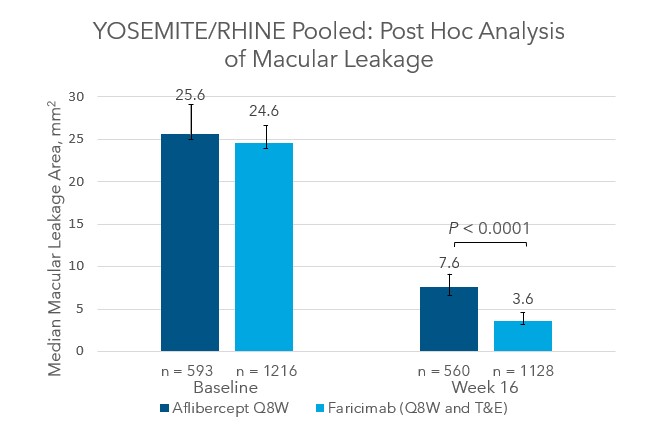
Figure 6. YOSEMITE and RHINE post hoc analysis of median macular leakage area of eyes treated with faricimab and aflibercept at baseline and after 16 weeks.15
Pradeep Prasad, MD, MBA: The PDS with ranibizumab is surgically implanted, and there is a black box warning for an increased risk of endophthalmitis associated with its use.16 Some patients prefer to avoid those potential complications. Other patients who have received the PDS with ranibizumab are really happy.
I also use steroid implants to treat DME. Typically, I start a patient with anti-VEGF then switch to combination anti-VEGF/anti-Ang-2 treatment. If that patient does not respond, then I move them to steroids. My first-line option is a dexamethasone implant unless there is a contraindication like uncontrolled glaucoma or an open posterior capsule. In some patients who have needed ongoing dexamethasone implants, I have tried using a fluocinolone implant. There is an ongoing debate about whether we should be using steroids earlier in the disease course. There is an inflammatory component to diabetic eye disease, and so it makes sense to use medications that target inflammatory mediators involved in this disease.
For some patients, laser is a treatment option—for example, a patient with a microaneurysm with surrounding exudates. The microaneurysm is well outside the fovea center, but edema is creeping into the central subfield and causing visual distortion or blurred vision. For those patients, laser can be very effective and can decrease treatment burden. I do not do a whole lot of laser, but it may make a lot of sense for certain patients.
Sharon Solomon, MD: I will add that I still manage women who have developed DME during pregnancy with focal laser. We do not know the effects of intravitreal anti-VEGF therapy in a pregnant mother and the risk to an unborn child. I certainly have patients for whom I would do an old-fashioned 30-degree fluorescein angiogram and then treat the leaking microaneurysms with focal laser. Those patients have done well.
With respect to peripheral disease, patients typically have a history of not having reliable follow-up. In addition, if the presentation is in an acute phase of retinal neovascularization or if the patient has a vitreous hemorrhage, in these cases, I will treat with anti-VEGF and follow that up with a panretinal laser. I think this yields a more durable regression of the disease.
Pradeep Prasad, MD, MBA: One other point I would like to make is that it is critical for us to be in good communication with the folks who are involved in taking care of our patients with diabetes, including primary care physicians and endocrinologists. Prevention is so much better than treating end-stage disease.
Benefits of Collaborative Care
Charles Vega, MD: I feel like I might be the Pollyanna of the group, because when it comes to patients presenting in acute situations in primary care, no group is more responsive than eye care specialists when it comes to a patient coming in with an ocular emergency. Ophthalmologists always seem to find a way to be able to fit that patient into their clinic schedule, without the patient having to go to the emergency department (ED). This is a huge help, particularly in the last few years with COVID. The ED is always slammed. It is very heartening to know that eye care specialists are willing to see patients acutely if the patient is having eye pain or sudden vision loss.
Regarding the treatment of diabetes, we’ve come a long way with more advanced therapeutics. Agents like GLP-1 agonists and SGLT2 inhibitors have really taken over, and even Medicaid and Medicare patients can get some of those agents covered. They may not directly impact eye health like they do cardiovascular and kidney health, but they are a lot more tolerable compared with older anti-diabetes drugs. These agents also promote weight reduction which also lowers A1c. We do not know if these agents have a more direct benefit on retinopathy; the clinical trials were never geared towards studying that outcome directly. I am hopeful because I see better management of diabetes that can counterbalance the growing wealth disparity and lack of access. We have better medications now; we just need to get them to the people who need them.
References
- Cheung N, Mitchell P, Wong TY. Diabetic retinopathy. Lancet. 2010;376(9735):124-136.
- Varma R, Bressler NM, Doan QV, et al. Prevalence of and risk factors for diabetic macular edema in the United States. JAMA Ophthalmol. 2014;132(11):1334-1340.
- Coney JM, Scott AW. Racial disparities in the screening and treatment of diabetic retinopathy. J Natl Med Assoc. 2022;114(2):171-181.
- Bressler NM, Varma R, Doan QV, et al. Underuse of the health care system by persons with diabetes mellitus and diabetic macular edema in the United States. JAMA Ophthalmol. 2014;132(2):168-173.
- Mora N, Kempen JH, Sobrin L. Diabetic retinopathy in Hispanics: a perspective on disease burden. Am J Ophthalmol. 2018;196:xviii-xxiv.
- Malhotra NA, Greenlee TE, Iyer AI, et al. Racial, ethnic, and insurance-based disparities upon initiation of anti-vascular endothelial growth factor therapy for diabetic macular edema in the US. Ophthalmology. 2021;128(10):1438-1447.
- Centers for Disease Control and Prevention. National Diabetes Statistics Report. https://www.cdc.gov/diabetes/data/statistics-report/index.html
- Solomon SD, Chew E, Duh EJ, et al. Diabetic retinopathy: a position statement by the American Diabetes Association. Diabetes Care. 2017;40(3):412-418.
- Cohen S, Murphy MLM, Prather AA. Ten surprising facts about stressful life events and disease risk. Annu Rev Psychol. 2019;70:577-597.
- Geronimus AT, Hicken M, Keene D, Bound J. "Weathering" and age patterns of allostatic load scores among blacks and whites in the United States. Am J Public Health. 2006;96(5):826-833.
- Tao Z, Clements NK. Transgenerational trauma and health inequity today. AMA J Ethics. 2021;23(6):E437-439.
- US Preventive Services Task Force; Davidson KW, Barry MJ, et al. Screening for prediabetes and type 2 diabetes: US Preventive Services Task Force recommendation statement. JAMA. 2021;326(8):736-743.
- National Association of Community Health Centers. Accessed May 26, 2023. https://www.nachc.org/
- Baumal C, Wells JA, Danzig CJ, et al. Efficacy, durability, and safety of faricimab in diabetic macular edema (DME): 2-year results from YOSEMITE and RHINE. Presented at the American Academy of Ophthalmology Annual Meeting; Chicago, IL; September 30-October 3, 2022.
- Goldberg R, Kolomeyer A, Nudleman E, et al. Faricimab reduces macular leakage vs aflibercept in patients with DME. Presented at ARVO 2023; New Orleans, LA; April 23-27, 2023.
- Ranibizumab injection for intravitreal use via SUSVIMO ocular implant. Package insert. Genentech, Inc.; 2022.










Facebook Comments