ReachMD
Be part of the knowledge.™Device That Closes Defect in Babies’ Hearts Is Safe, Effective
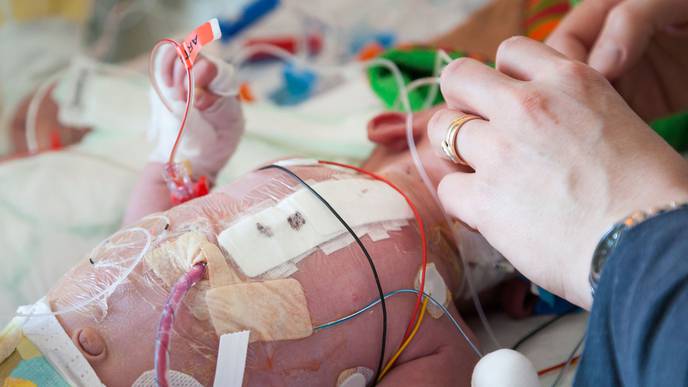
A device pioneered in partnership with Cedars-Sinai to close the most common heart defect in premature babies is safe and effective, according to the results of a clinical trial recently published in the peer-reviewed Journal of Perinatology.
“This device has changed the lives of many of our tiniest patients and their families, who struggled with a very common but serious condition,” said Evan Zahn, MD, co-director of the Guerin Family Congenital Heart Program and director of the Division of Pediatric Cardiology at Cedars-Sinai Guerin Children’s and the Smidt Heart Institute. “It has meant fewer heart surgeries or medications that can cause dangerous side effects.”
Zahn was the first surgeon to implant the pea-sized mesh device, known as the Amplatzer Piccolo Occluder, to close a hole in the heart of a baby born with severe prematurity. The device was approved by the U.S. Food and Drug Administration in 2019.
Piccolo implantation is a minimally invasive approach designed to address patent ductus arteriosus (PDA), which is the most common heart defect seen in premature infants.
The ductus arteriosus is a blood vessel between the heart’s two major arteries that allows a fetus’ blood to bypass its lungs while it relies on its mother for oxygenated blood. Once a baby is born and needs to breathe on its own, the ductus arteriosus naturally closes. If it doesn’t, it can cause extra blood to flow into the baby’s pulmonary arteries and can contribute to severe lung disease, bleeding in the brain, problems with the bowels and even death.
Medication therapy for PDA can cause side effects, such as kidney failure and bleeding, and heart surgery to close the PDA can lead to long-term health issues.
To implant the Amplatzer Piccolo Occluder, a physician guides a catheter through a vein in the leg to the heart and closes the hole. The procedure takes a few minutes.
For this study, investigators looked into PDA-related health outcomes in 200 premature babies who were enrolled in the Piccolo clinical trial between June 2017 and February 2019. Nine children enrolled in the study died, but the deaths were determined to be unrelated to the device or procedure.
Imaging showed full PDA closure in the 33 patients for whom this data was available three years after they had the Piccolo device implanted.
Zahn and his team have since developed a technique to perform the procedure at the bedside, rather than in the catheterization laboratory, as was done during this clinical trial.
According to the study authors, “This offers the benefit of allowing experienced practitioners to continue to care for these patients in the units that know them best without the need for transport.”
Funding: The study was funded by Abbott Structural Heart, the company that makes the Amplatzer Piccolo Occluder.
Conflicts of interest: Several study authors reported conflicts of interest. Brian H. Morray is a consultant and proctor for Abbott Structural Heart. Shyam K. Sathanandam: proctor/consultant for Abbott. Thomas Forbes: proctor/consultant for Abbott, Edwards, AcuNav/Biosence Webster, B. Braun Medical, Siemens, Medtronic. Matthew Gillespie: proctor/consultant for Abbott, Medtronic, W.L. Gore & Assoc. Darren Berman: proctor/consultant for Abbott, Edwards, Medtronic. Aimee K. Armstrong: proctor/consultant for Abbott, Edwards, Medtronic, Cook Medical. S. Shahanavaz: proctor for Abbott, Medtronic, Edwards. Thomas Jones: research grant; proctor/consultant for Abbott, Edwards, Medtronic, W.L. Gore & Assoc.. Toby Rockefeller: proctor for Abbott. Henri Justino: proctor/consultant for Abbott, Baylis Medtech, Chiesi USA, Edwards Lifesciences, Medtronic; Clinical Trial Executive Committee for Janssen Pharmaceutical; co-founder of PolyVascular; scientific advisory board for Pediastent. David Nykanen: proctor for Abbott; consultant and independent data reviewer for W.L. Gore & Assoc.; expert witness for Glaxo Smith Kline. Courtney Weiler: full-time employee at Abbott. Dan Gutfinger: full-time employee at Abbott. Evan M. Zahn: consultant/proctor for Abbott, Edwards, Medtronic, National PI ADO II AS IDE Trial and Alterra/S3.
Read more on the Cedars-Sinai Blog:Making Strides in Treating the Smallest Heart Patients
Facebook Comments