ReachMD
Be part of the knowledge.™Bausch + Lomb Launches Dry Eye Drug Miebo in the United States - Eyewire+
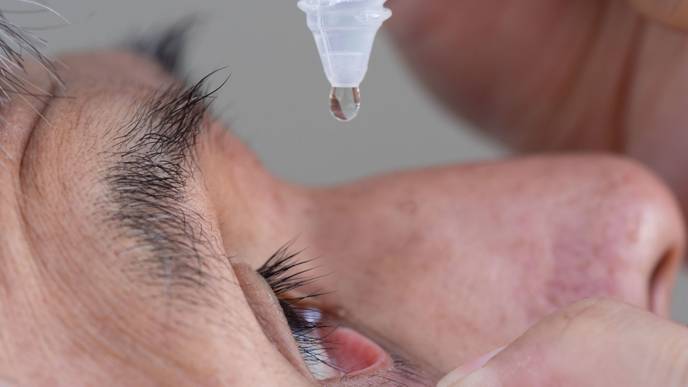
Bausch + Lomb announced the US commercial launch of Miebo (perfluorohexyloctane ophthalmic solution) for the treatment of the signs and symptoms of dry eye disease (DED).
In May, Bausch + Lomb and Novaliq received FDA for Miebo as the first and only FDA-approved treatment for DED that directly targets tear evaporation. Miebo is a single ingredient, water-, preservative- and steroid-free prescription eye drop administered as a single drop into each eye and is given four times a day. It is designed to reduce tear evaporation at the ocular surface.
“More and more Americans are suffering from dry eye disease, due in large part to today’s multi-screen lifestyles,” Andrew Stewart, president, Global Pharmaceuticals and International Consumer, Bausch + Lomb, said in a company news release. “With Miebo, eye care professionals now have a prescription eye drop that directly addresses tear evaporation, the leading cause of dry eye disease.”
To view EyewireTV's coverage of the FDA approval of Miebo, including an exclusive interview with Mr. Stewart, click here.
The FDA approval of Miebo was based on results from two 57-day, multicenter, randomized, double-masked, saline-controlled studies, GOBI and MOJAVE, which enrolled a total of 1,217 patients with a history of DED and clinical signs of MGD, a major cause of development and disease progression. An estimated 86% of people with DED have excessive tear evaporation whereby MGD is the major contributor.
In the GOBI and MOJAVE, Miebo met both primary sign and symptom efficacy endpoints. The two primary endpoints were change from baseline at week 8 (day 57 ± 2) in total corneal fluorescein staining (tCFS) and eye dryness Visual Analog Scale (VAS) score. Patients experienced relief of symptoms as early as day 15 and through day 57 with statistically significant reduction in VAS eye dryness score favoring Miebo observed in both studies. Additionally, at days 15 and day 57, a significant reduction in tCFS favoring MIEBO was observed in both studies.
For more information on Miebo, visit www.MIEBO.com.
Facebook Comments